We also know that they are made up of smaller particles such as quarks and leptons.Īfter the Big Bang, 13.7 billion years ago, our very hot universe began to expand and become cooler, allowing the formation of quarks and leptons. But now we know that atoms are made up of three types of particles: protons, neutrons, and electrons. The term “atom” comes from the Greek word Atomus, which means “indivisible.” At that time, the Greeks believed that the atom was the smallest thing in the universe and could not be divided. The atom is the smallest building stone or the smallest part of a chemical element that retains the chemical properties of that element, and thus it is considered as the basic unit of matter that controls the structure of the elements. Let’s start our journey from the atom to understand what it means. Other particles are produced from nuclear reactions, but they are unstable. Protons and neutrons are made up of smaller particles called quarks. Subatomic particles are particles much smaller than atoms, such as protons, electrons, and neutrons that make up the atom. The term particle always refers to objects of small sizes, and the most important types of particles are atomic and subatomic particles, each of which has different properties. In the physical sciences, a particle is a small localized object to which can be ascribed several physical or chemical properties, such as volume, density, or mass. What are The Three Types of Subatomic Particles?īefore we get to know subatomic particles, let’s first get to know the particles in chemistry and physics.Figure 2.4 "Rutherford’s Metal-Foil Experiments" shows how this model explains the experimental results. Rutherford called his description the “planetary model” of the atom. As a result, an atom consists largely of empty space. Most of the mass of an atom is in the nucleus, while the orbiting electrons account for an atom’s size. 
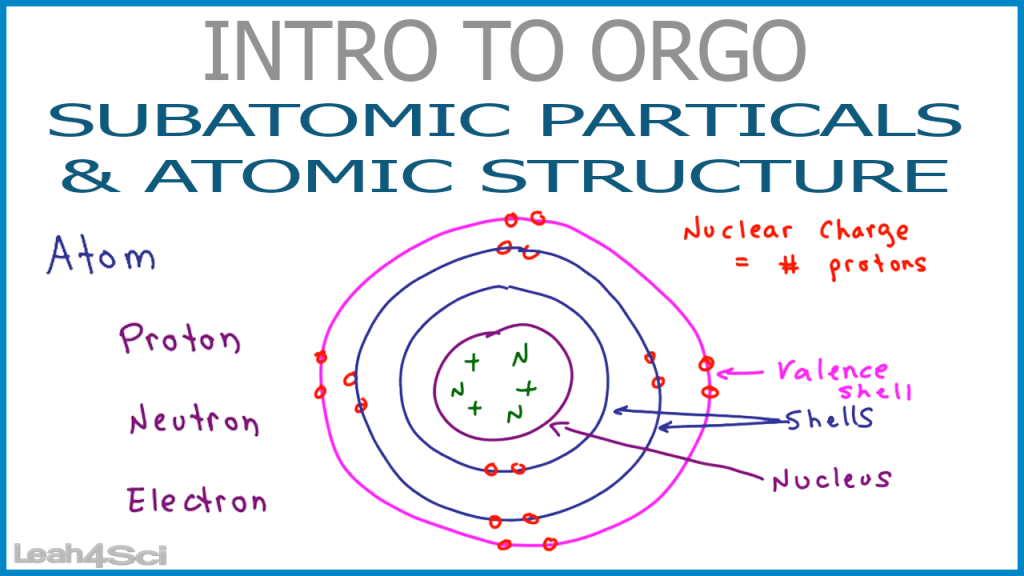
Electrons are outside the nucleus and orbit about it because they are attracted to the positive charge in the nucleus. Protons and neutrons are concentrated in a central region he called the nucleus The central part of an atom that contains protons and neutrons. 
Rutherford proposed the following model to explain these experimental results. Rutherford once said, “It was almost as incredible as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you.” Some were even deflected back toward the source. Most of the particles traveled straight through the foil, but some alpha particles were deflected off to one side. Table 2.4 "Properties of the Subatomic Particles" lists some of their important characteristics and the symbols used to represent each particle.įigure 2.3 The Geiger-Marsden Experimental SetupĮxperiments using this setup were used to investigate the structure of atoms. We understand now that all atoms can be broken down into subatomic particles: protons, neutrons, and electrons. Evidence produced in 1932 established the existence of the neutron A subatomic particle with no electric charge., a particle with about the same mass as a proton but with no electrical charge. Because opposite charges attract each other (while like charges repel each other), protons attract electrons (and vice versa).įinally, additional experiments pointed to the existence of a third particle. Although still incredibly small, the mass of a proton is 1.673 × 10 −27 kg, which is almost 2,000 times greater than the mass of an electron. Another major difference between a proton and an electron is mass. 
has the same amount of charge as an electron, but its charge is positive, not negative. A proton A subatomic particle with a positive charge. Experiments with magnetic fields showed that the electron has a negative electrical charge.īy 1920, experimental evidence indicated the existence of a second particle. It is an extremely tiny particle, with a mass of about 9.109 × 10 −31 kg. The first subatomic particle was identified in 1897 and called the electron A subatomic particle with a negative electric charge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
